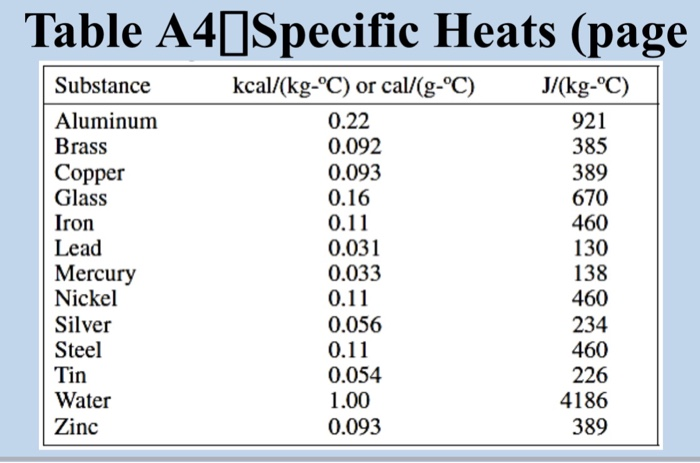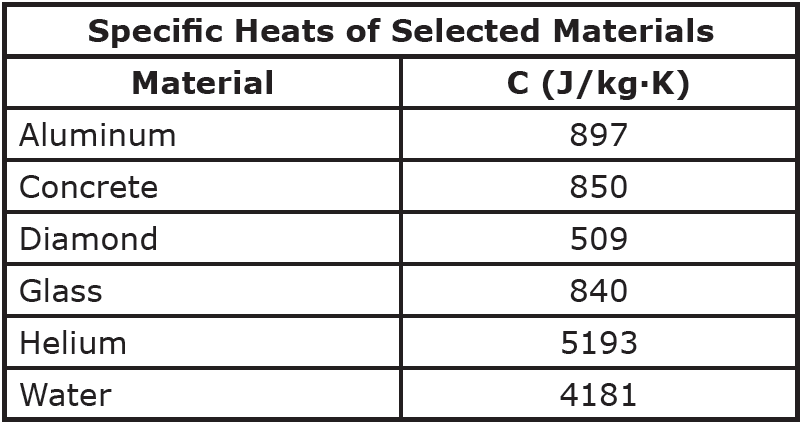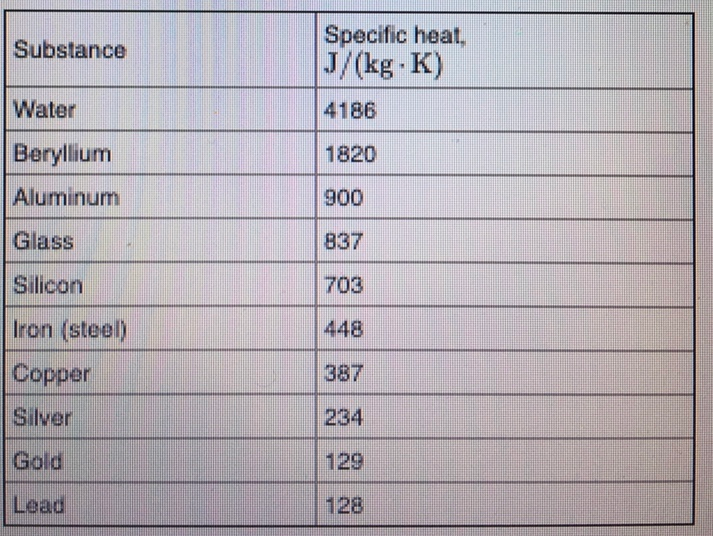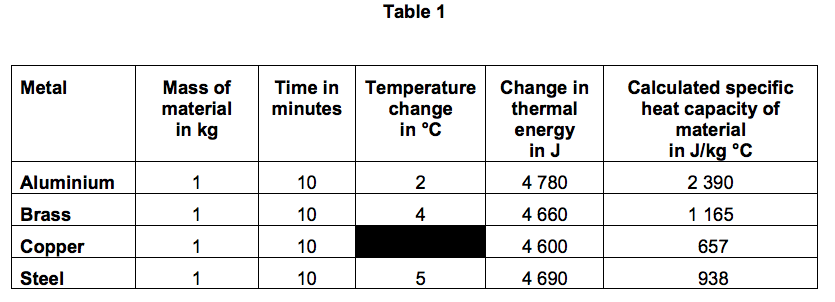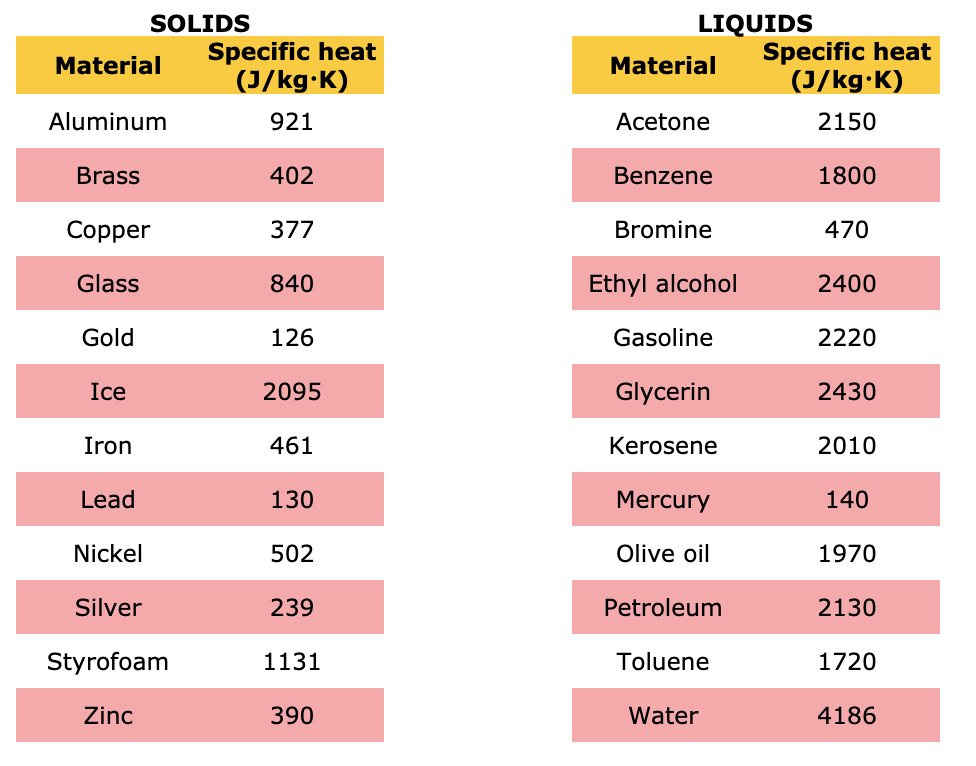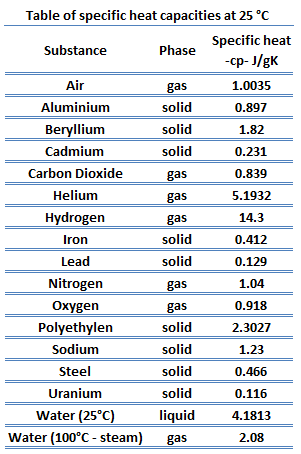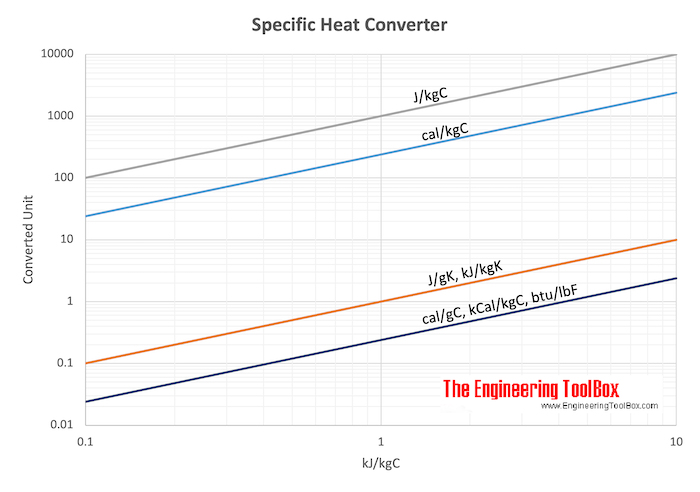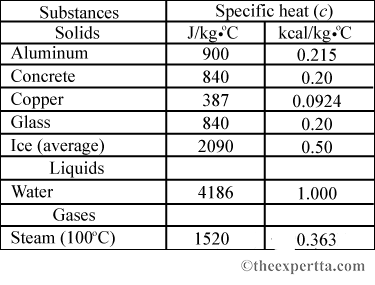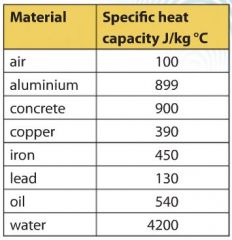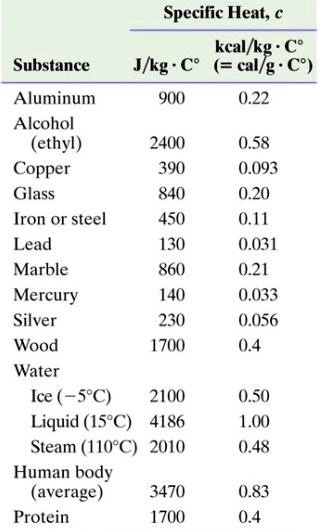
SOLVED: Specific Heat; kcal/kg J/kg Co (= callg C? 900 0.22 Substance Aluminum Alcohol (ethyl) 2400 Copper 390 Glass 840 Iron or steel 450 Lead 130 Marble 860 Mercury 140 Silver 230

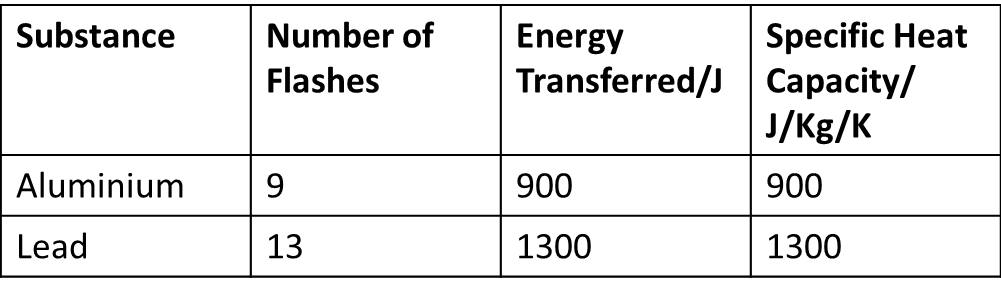
Cheat calculations Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online
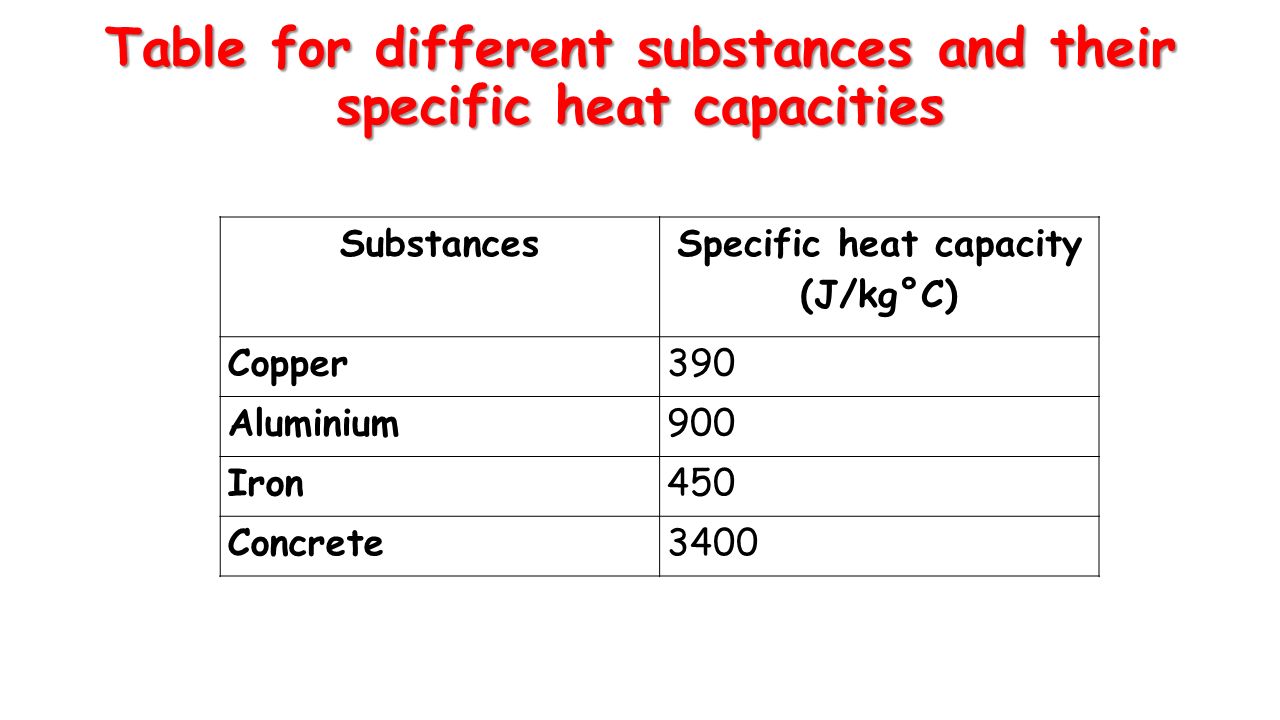
Learning Outcomes: Rearranging equation for Specific Heat Capacity Topic Equation for Specific Heat Capacity Target Audience: G & T Teacher instructions. - ppt download
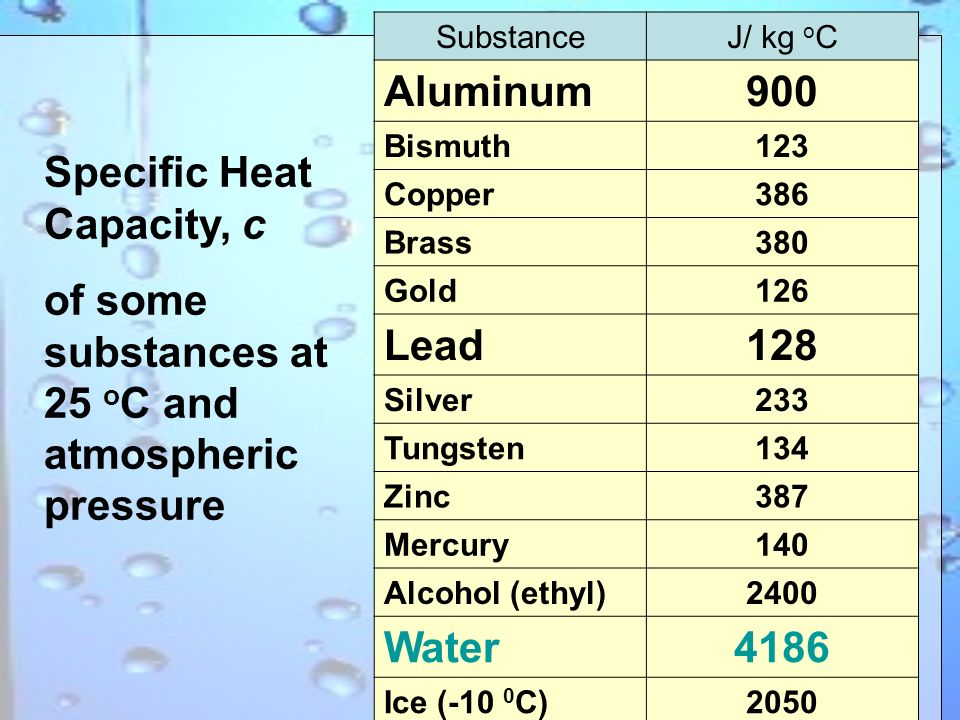
3b Thermodynamics Specific heat capacity, c Latent heat capacity, L Change of phase, evaporation First law of thermodynamics, - ppt download